Bi-specific Lipid Nanoparticles for Boosting Antitumor Immunity in Glioblastoma
SHORT DESCRIPTION
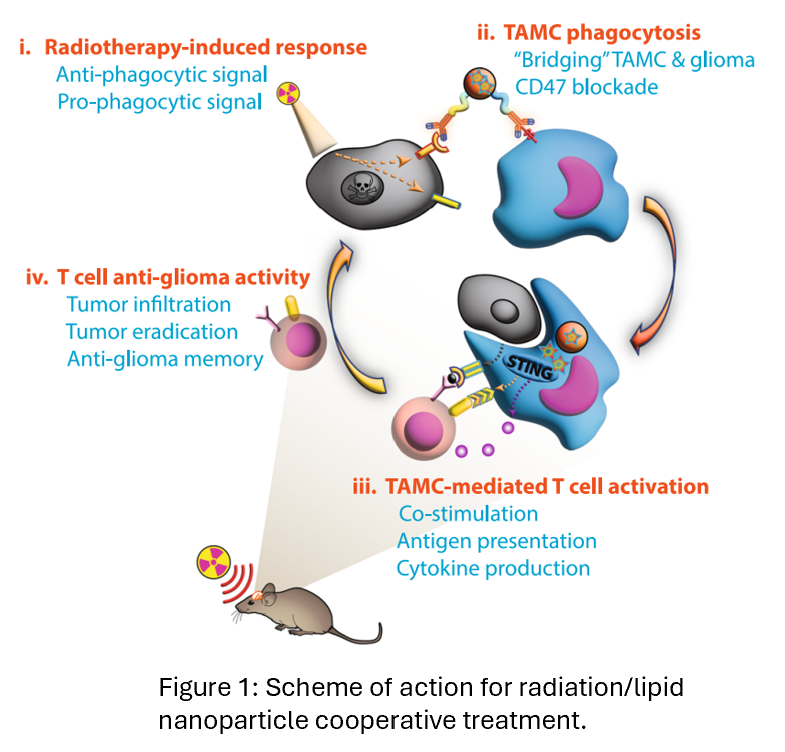
A bispecific lipid nanoparticle that bridges tumor-associated myeloid cells (TAMCs) and glioma cells by dual checkpoint blockade and simultaneously reprograms TAMCs into antitumor effectors via targeted delivery of a STING agonist.
INVENTORS
- Maciej Lesniak*
- Northwestern University Feinberg School of Medicine, Department of Neurological Surgery
- Peng Zhang
* Principal Investigator |
NU 2022-151
IP STATUS
US Patent filed (19/122,639).
DEVELOPMENT STAGE
TRL-5 Prototype Validated in Relevant Environment: Preclinical murine studies demonstrate significant antitumor efficacy when combined with radiotherapy.
|
 BACKGROUND
BACKGROUND
Glioblastoma is the most common and highly aggressive primary malignant brain tumor, accounting for roughly half of all malignant brain tumors and about 12,000 new cases annually in the U.S. Standard-of-care remains maximal safe surgical resection followed by radiotherapy and temozolomide-based chemotherapy; however, with a 5‑year survival around 6–7% and median overall survival at about 12–18 months, glioblastoma remains the leading cause of brain tumor–related death and is responsible for the majority of deaths among primary brain tumor patients. Current glioblastoma treatments, including radiation therapy, often trigger immune resistance and promote tumor evasion through immune checkpoint upregulation, contributing to near-universal recurrence, and face challenges such as high treatment costs and limited efficacy, underscoring a critical need for new and more effective treatment strategies.
ABSTRACT
Northwestern scientists developed a bispecific lipid nanoparticle (B-LNP) 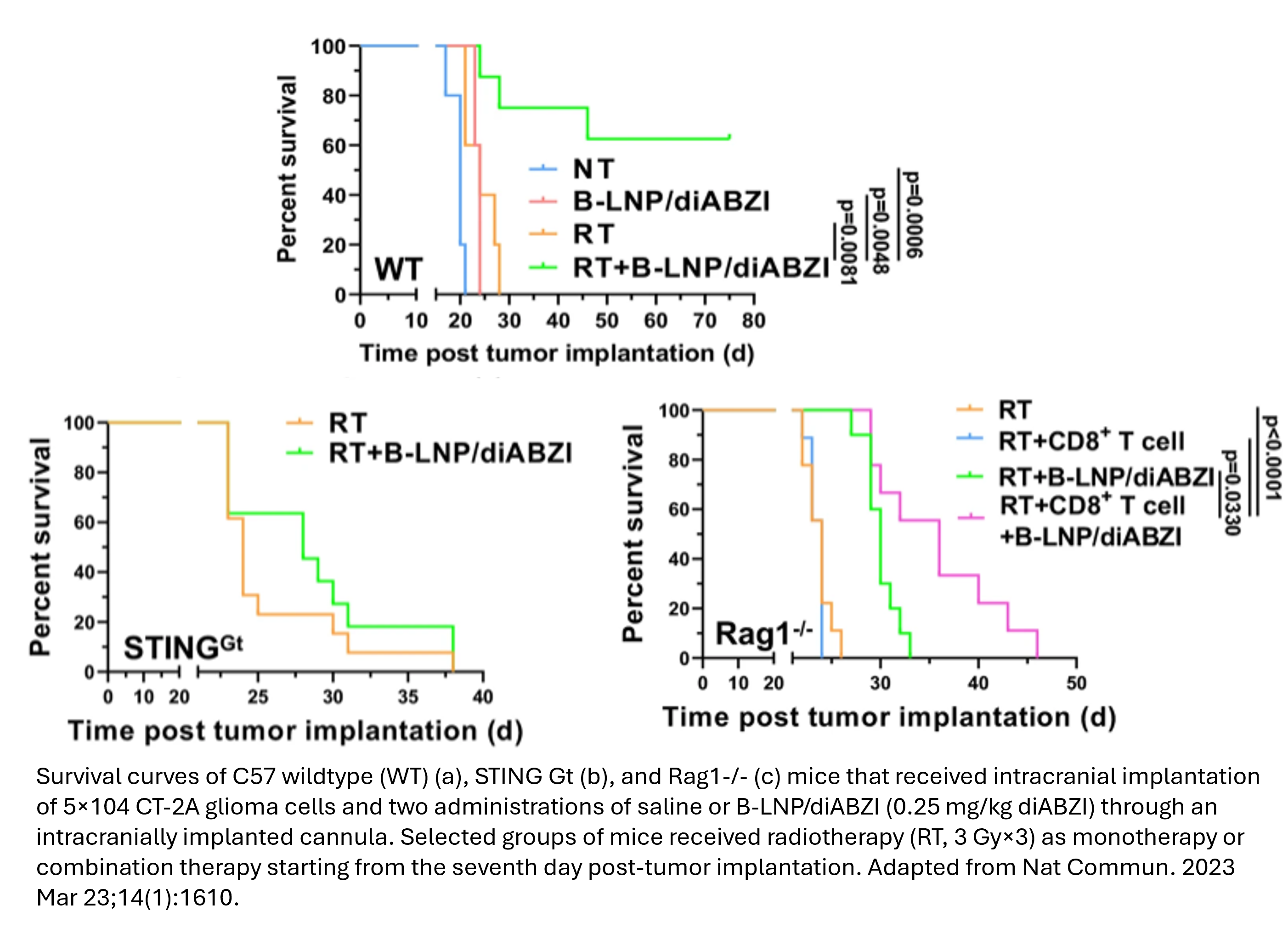 that targets tumor-associated myeloid cells (TAMCs) and glioma cells via dual ligation of CD47 and PD-L1. The nanoparticle blocks these immune checkpoints and delivers a STING agonist (diABZI) to reprogram TAMCs into antitumor effectors. In preclinical murine models, the B-LNP, when combined with radiotherapy, increased phagocytosis and T cell activation leading to significant tumor regression.
that targets tumor-associated myeloid cells (TAMCs) and glioma cells via dual ligation of CD47 and PD-L1. The nanoparticle blocks these immune checkpoints and delivers a STING agonist (diABZI) to reprogram TAMCs into antitumor effectors. In preclinical murine models, the B-LNP, when combined with radiotherapy, increased phagocytosis and T cell activation leading to significant tumor regression.
APPLICATIONS
- Glioblastoma treatment: Enhances antitumor immune responses in brain tumors.
- Combination therapy with radiotherapy: Boosts radiation efficacy through immune reprogramming.
- Targeted immunotherapy: Delivers STING agonists directly to tumor-associated myeloid cells.
- Nanoparticle-mediated drug delivery: Improves precision and safety in immune checkpoint blockade.
ADVANTAGES
- Enhances immune cell phagocytosis: Blocks dual immune checkpoints for focused antitumor activity.
- Synergizes with radiotherapy: Amplifies tumor regression when used with standard treatments.
- Promotes durable responses: Reprograms myeloid cells to sustain long-term antitumor immunity.
- Precision delivery: Targets specific immune cells, reducing off-target effects and improving safety.
PUBLICATIONS
CATEGORY/INDUSTRY PIPELINE
 Healthcare Devices, Tools & IT; Therapeutics
Healthcare Devices, Tools & IT; Therapeutics
KEYWORDS
Cancer, oncology, glioblastoma, brain cancer, bispecific lipid nanoparticles, immunotherapy, tumor-associated myeloid cells, STING agonist, immune checkpoint blockade, radiotherapy, nanoparticle, drug delivery, targeted therapy
Patent Information:
| Title |
App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |
Categories:
Life Sciences > Therapeutics
Keywords:
Brain cancer
Cancer/Oncology
Drug delivery
Immunotherapy
Nanoparticle
Nanotechnology
Targeted therapy
Therapeutics