Responsive Probiotic Therapy for Local Detection and Treatment of IBD Flares
Case ID:
Prindle 2023-131
SHORT DESCRIPTION
An engineered probiotic that senses intestinal inflammation via calprotectin and locally releases therapeutic antibodies in the gastrointestinal tract only during inflammatory flares.
INVENTORS
- Arthur Prindle*
- McCormick School of Engineering, Department of Chemical and Biological Engineering
- Jonathan Xia
* Principal Investigator |
NU 2023-131, NU 2024-047
IP STATUS
Multiple US Patents pending (18/775,012; 19/198,291)
DEVELOPMENT STAGE
TRL-5 Prototype Validated in Relevant Environment: Calprotectin sensor performance has been confirmed using a DSS-induced colitis murine model.
|
BACKGROUND
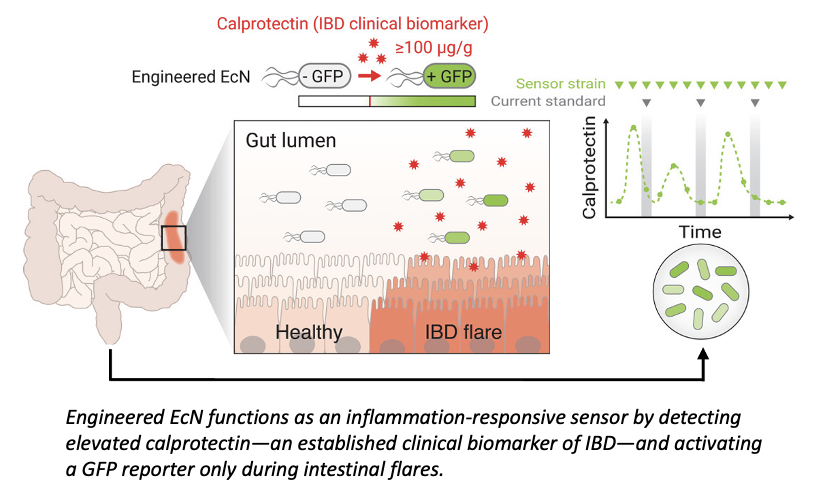 Inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, is a chronic autoimmune condition marked by recurrent inflammation of the gastrointestinal (GI) tract, requiring long‑term management, which is associated with substantial morbidity, impaired quality of life, increased colorectal cancer risk, and significant healthcare costs. Globally, IBD affects an estimated 4.9 million people, with prevalence rising. While effective therapies exist, they are typically delivered systemically and can cause significant side effects due to prolonged immunosuppression. At the same time, disease monitoring relies on invasive or slow diagnostic methods, which can delay timely intervention during inflammatory flares. There is a critical need for therapeutic approaches that both detect intestinal inflammation noninvasively and deliver treatment locally, only when disease activity is present.
Inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, is a chronic autoimmune condition marked by recurrent inflammation of the gastrointestinal (GI) tract, requiring long‑term management, which is associated with substantial morbidity, impaired quality of life, increased colorectal cancer risk, and significant healthcare costs. Globally, IBD affects an estimated 4.9 million people, with prevalence rising. While effective therapies exist, they are typically delivered systemically and can cause significant side effects due to prolonged immunosuppression. At the same time, disease monitoring relies on invasive or slow diagnostic methods, which can delay timely intervention during inflammatory flares. There is a critical need for therapeutic approaches that both detect intestinal inflammation noninvasively and deliver treatment locally, only when disease activity is present.
ABSTRACT
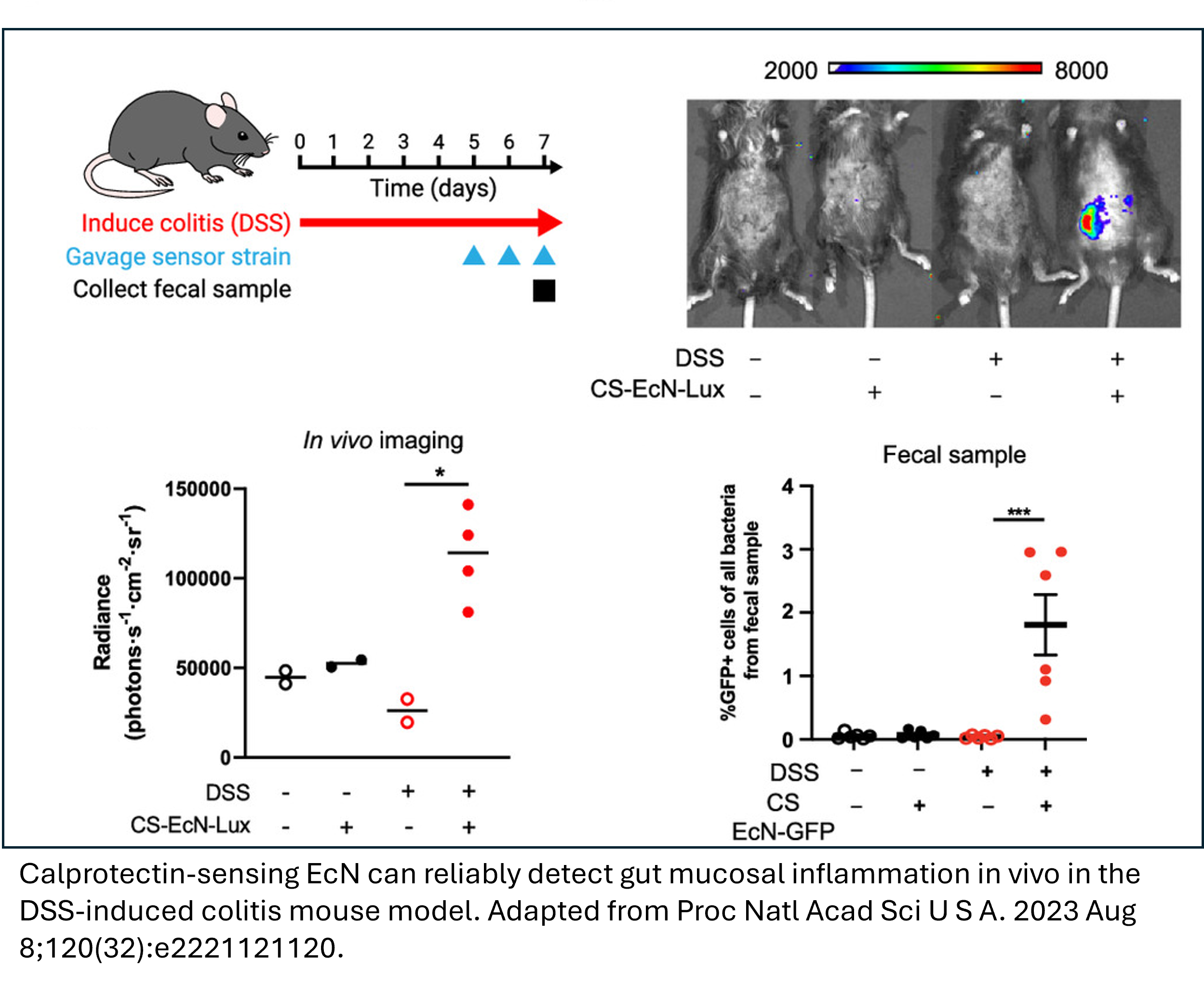 Northwestern researchers have developed an engineered probiotic capable of detecting markers indicative of active inflammation in the GI tract and releasing therapeutic antibodies in response for treatment of IBD. This invention integrates synthetic biology with microbial diagnostics. E. Coli Nissle 1917 (EcN)—a probiotic with established safety in humans—was engineered to produce and selectively release therapeutic antibodies in the GI tract only in the presence of calprotectin, the clinical gold standard biomarker of IBD. In both in vitro settings and a DSS-induced murine colitis model, the probiotic showed high sensitivity and specificity to inflammation. The platform distinguished active IBD from remission by quantifying reporter signals that track with clinical calprotectin levels. For disease management, the engineered probiotic can subsequently release a therapeutic antibody, such as anti-TNF-α.
Northwestern researchers have developed an engineered probiotic capable of detecting markers indicative of active inflammation in the GI tract and releasing therapeutic antibodies in response for treatment of IBD. This invention integrates synthetic biology with microbial diagnostics. E. Coli Nissle 1917 (EcN)—a probiotic with established safety in humans—was engineered to produce and selectively release therapeutic antibodies in the GI tract only in the presence of calprotectin, the clinical gold standard biomarker of IBD. In both in vitro settings and a DSS-induced murine colitis model, the probiotic showed high sensitivity and specificity to inflammation. The platform distinguished active IBD from remission by quantifying reporter signals that track with clinical calprotectin levels. For disease management, the engineered probiotic can subsequently release a therapeutic antibody, such as anti-TNF-α.
APPLICATIONS
- IBD Disease Activity Monitoring: Enables noninvasive tracking of gut inflammation.
- Early Intervention Strategies: Supports timely identification of IBD flares to prevent complications.
- IDB Disease Management: Enables precise, inflammation‑responsive medication delivery within the gastrointestinal tract.
ADVANTAGES
- Noninvasive and rapid: Eliminates the need for colonoscopies while providing fast results.
- Highly sensitive and specific: Reduces systemic immunosuppression from IV infusions by enabling local, calprotectin-induced delivery of antibody therapeutics.
- Cost-effective: Reduces overall diagnostic expenses by streamlining monitoring processes.
- Real-time disease tracking: Supports immediate clinical decisions and proactive patient care.
PUBLICATIONS
 KEYWORDS
KEYWORDS
Inflammation, immunology, IBD, calprotectin, engineered probiotics, non-invasive monitoring, biosensor, synthetic biology, gut inflammation, diagnostics, autoimmunity, biologic, cell therapy, therapeutics
Patent Information:
| Title |
App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |
Categories:
Life Sciences > Therapeutics
Keywords:
Autoimmune disease
Biologic
Cell therapy
Diagnostics
Gastrointestinal and Hepatic disease
IBD - Inflammatory bowel disease
Immunology
Immunotherapy
Inflammation
Synthetic biology
Therapeutics